Medical device regulation in europe
Home » Doctor Visit » Medical device regulation in europeMedical device regulation in europe
Medical Device Regulation In Europe. Please click here the european commission medical device website and for more information on medical device registration in europe. Usually it is the same person who the management representative as defined by iso 13485. The regulatory landscape for medical devices in europe is undergoing some major changes. The final texts of the new european medical devices regulation (mdr) and ivd regulation (ivdr) have been published in the official journal of the european union.
 Faq On The European Medical Device Regulation - B Medical Systems (Fr) From bmedicalsystems.com
Faq On The European Medical Device Regulation - B Medical Systems (Fr) From bmedicalsystems.com
Over the past decade, a number of scandals emerged regarding patients harmed as a result of treatment with approved medical devices. The eu mdr is the european union medical device regulation 2017/745 released in 2017 by the european parliament and the council of the european union. On may 26, 2021, the european union (eu) implemented regulation (eu) 2017/745, the “medical device regulation,” or mdr. 1) the first thing you need is to get a qualified prrc (person responsible for regulatory compliance). The new regulation of medical devices aims to establish the path for medical device manufacturers to enter the european market. Effective may 26th 2021, the european union medical device regulation (mdr) governing market access to the european market, is the most significant regulatory change in over 20 years.
After its first year in existence, the medtech community’s initial concerns with the mdr’s requirements have now become a matter of increasing frustration for.
There are over 27,000 medtech companies in europe, 95% of which have revenues of less than €50m, and about half a million product. The european market and regulatory change. The new regulation of medical devices aims to establish the path for medical device manufacturers to enter the european market. Over the past decade, a number of scandals emerged regarding patients harmed as a result of treatment with approved medical devices. We have two different regulations: The medical device regulation (mdr), which was adopted in april 2017, changes the european legal framework for medical devices and introduces new principal and supportive responsibilities for ema and for national competent authorities in the assessment of certain categories of products.

The eu mdr represents the unified european regulatory requirements for medical devices that must be followed throughout europe to secure and maintain your ce mark. Regulation (eu) 2017/745 of 5 april 2017 on medical devices that has gone into effect on the 26th may 2021. 1) the first thing you need is to get a qualified prrc (person responsible for regulatory compliance). Published in may 2017, the new european medical device regulation (mdr) (regulation [eu] 2017/745) and in vitro diagnostics regulation (ivdr) (regulation [eu] 2017/746) is gradually replacing the medical devices directive and the implantable medical devices. The new regulation of medical devices aims to establish the path for medical device manufacturers to enter the european market.
 Source: hpra.ie
Source: hpra.ie
The european market and regulatory change. There are over 27,000 medtech companies in europe, 95% of which have revenues of less than €50m, and about half a million product. After its first year in existence, the medtech community’s initial concerns with the mdr’s requirements have now become a matter of increasing frustration for. Medical devices have been regulated in a coordinated fashion throughout europe since the 1990s. The european union medical device regulation (eu mdr) is a new directive that has fully superseded its predecessor, the mdd (medical devices directive).
 Source: tuv.com
Source: tuv.com
After its first year in existence, the medtech community’s initial concerns with the mdr’s requirements have now become a matter of increasing frustration for. Commission implementing regulation (eu) 2022/1107 of 4 july 2022 laying down common specifications for certain class d in vitro diagnostic medical devices in accordance with regulation (eu) 2017/746 of the european parliament and of the council. Over the past decade, a number of scandals emerged regarding patients harmed as a result of treatment with approved medical devices. The mdr, which replaces the medical. Medical devices have been regulated in a coordinated fashion throughout europe since the 1990s.
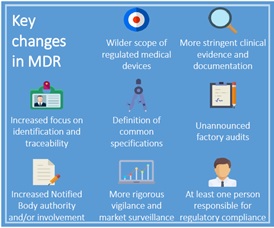 Source: catchtrial.com
Source: catchtrial.com
Valued at €110bn in 2017, the european market for medical devices represents around 30% of the global industry; To give you an idea: Effective may 26th 2021, the european union medical device regulation (mdr) governing market access to the european market, is the most significant regulatory change in over 20 years. Unlike the previous medical device directive, the mdr is directly applicable, without exception, in all european member states. The regulatory landscape for medical devices in europe is undergoing some major changes.
 Source: eurointervention.pcronline.com
Source: eurointervention.pcronline.com
On may 26, 2021, the european union (eu) implemented regulation (eu) 2017/745, the “medical device regulation,” or mdr. The mdr, which replaces the medical. The new regulation of medical devices aims to establish the path for medical device manufacturers to enter the european market. However, in may 2021, the uk’s medical device laws deviated from those of the eu, due to the adoption of new regulations. With the formal publication of guidance imminent, the proposals will give national regulators much more control and oversight of the medical devices industry—with adoption mandatory.
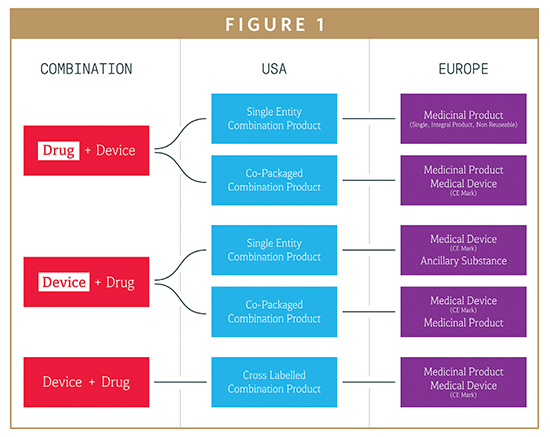 Source: drug-dev.com
Source: drug-dev.com
Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive 2001/83/ec, regulation (ec) no 178/2002 and regulation (ec) no 1223/2009 and repealing council directives 90/385/eec and 93/42/eec (text with eea relevance. Regulation (eu) 2017/745 of 5 april 2017 on medical devices that has gone into effect on the 26th may 2021. After its first year in existence, the medtech community’s initial concerns with the mdr’s requirements have now become a matter of increasing frustration for. We have two different regulations: To give you an idea:
 Source: mhc.ie
Source: mhc.ie
The mdr, which replaces the medical. Effective may 26th 2021, the european union medical device regulation (mdr) governing market access to the european market, is the most significant regulatory change in over 20 years. This new set of regulations will replace existing directives of the eu regulatory framework, medical devices directive (mdd) and active implantable medical devices (aimd) directive, to keep up with scientific advances in the medical industry, improve. Regulation (eu) 2017/745 of the european parliament and of the council of 5 april 2017 on medical devices, amending directive 2001/83/ec, regulation (ec) no 178/2002 and regulation (ec) no 1223/2009 and repealing council directives 90/385/eec and 93/42/eec (text with eea relevance. There are over 27,000 medtech companies in europe, 95% of which have revenues of less than €50m, and about half a million product.
 Source: tuvsud.com
Source: tuvsud.com
Entry into force commenced on may 25th 2017, marking the start of the transition period for manufacturers selling medical devices into europe. Unlike the previous medical device directive, the mdr is directly applicable, without exception, in all european member states. Published in may 2017, the new european medical device regulation (mdr) (regulation [eu] 2017/745) and in vitro diagnostics regulation (ivdr) (regulation [eu] 2017/746) is gradually replacing the medical devices directive and the implantable medical devices. A second c orrigendum to regulation (eu) 2017/745 was published in the official journal of the european union. Valued at €110bn in 2017, the european market for medical devices represents around 30% of the global industry;
 Source: emergobyul.com
Source: emergobyul.com
With the formal publication of guidance imminent, the proposals will give national regulators much more control and oversight of the medical devices industry—with adoption mandatory. Medical devices have been regulated in a coordinated fashion throughout europe since the 1990s. The eu mdr represents the unified european regulatory requirements for medical devices that must be followed throughout europe to secure and maintain your ce mark. Regulation (eu) 2017/745 of 5 april 2017 on medical devices that has gone into effect on the 26th may 2021. This new set of regulations will replace existing directives of the eu regulatory framework, medical devices directive (mdd) and active implantable medical devices (aimd) directive, to keep up with scientific advances in the medical industry, improve.
 Source: medicaldeviceacademy.com
Source: medicaldeviceacademy.com
- the first thing you need is to get a qualified prrc (person responsible for regulatory compliance). With the formal publication of guidance imminent, the proposals will give national regulators much more control and oversight of the medical devices industry—with adoption mandatory. Please click here the european commission medical device website and for more information on medical device registration in europe. We have two different regulations: The regulatory landscape for medical devices in europe is undergoing some major changes.
 Source: globalregulatorypartners.com
Source: globalregulatorypartners.com
The european market and regulatory change. Unlike the previous medical device directive, the mdr is directly applicable, without exception, in all european member states. Commission implementing regulation (eu) 2022/1107 of 4 july 2022 laying down common specifications for certain class d in vitro diagnostic medical devices in accordance with regulation (eu) 2017/746 of the european parliament and of the council. On may 26, 2021, the european union (eu) implemented regulation (eu) 2017/745, the “medical device regulation,” or mdr. 1) the first thing you need is to get a qualified prrc (person responsible for regulatory compliance).
 Source: emergobyul.com
Source: emergobyul.com
The eu mdr is the european union medical device regulation 2017/745 released in 2017 by the european parliament and the council of the european union. Commission implementing regulation (eu) 2021/2226 of 14. The u.s., the market leader, accounts for about 42%. However, in may 2021, the uk’s medical device laws deviated from those of the eu, due to the adoption of new regulations. The eu mdr represents the unified european regulatory requirements for medical devices that must be followed throughout europe to secure and maintain your ce mark.
 Source: emergobyul.com
Source: emergobyul.com
The eu mdr is the european union medical device regulation 2017/745 released in 2017 by the european parliament and the council of the european union. The european union medical device regulation (eu mdr) is a new directive that has fully superseded its predecessor, the mdd (medical devices directive). The european market and regulatory change. 26 may 2022, the regulation (eu) 2017/746 on in vitro diagnostic medical devices is applicable within the eu. The final texts of the new european medical devices regulation (mdr) and ivd regulation (ivdr) have been published in the official journal of the european union.

A second c orrigendum to regulation (eu) 2017/745 was published in the official journal of the european union. The mdr, which replaces the medical. The european market and regulatory change. The final texts of the new european medical devices regulation (mdr) and ivd regulation (ivdr) have been published in the official journal of the european union. However, in may 2021, the uk’s medical device laws deviated from those of the eu, due to the adoption of new regulations.
 Source: bmedicalsystems.com
Source: bmedicalsystems.com
The medical device regulation (mdr), which was adopted in april 2017, changes the european legal framework for medical devices and introduces new principal and supportive responsibilities for ema and for national competent authorities in the assessment of certain categories of products. Unlike the previous medical device directive, the mdr is directly applicable, without exception, in all european member states. Effective may 26th 2021, the european union medical device regulation (mdr) governing market access to the european market, is the most significant regulatory change in over 20 years. The regulation entered into force in may 2017 and had a. The u.s., the market leader, accounts for about 42%.
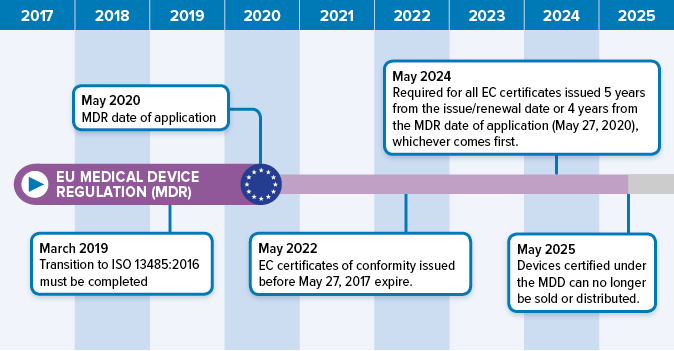 Source: orielstat.com
Source: orielstat.com
The european medical device regulation (mdr) is a new set of regulations that will become effective may 2020. Unlike the previous medical device directive, the mdr is directly applicable, without exception, in all european member states. The european medical device regulation (mdr) is a new set of regulations that will become effective may 2020. To give you an idea: Effective may 26th 2021, the european union medical device regulation (mdr) governing market access to the european market, is the most significant regulatory change in over 20 years.
 Source: magazine.zhermack.com
Source: magazine.zhermack.com
To give you an idea: To give you an idea: After its first year in existence, the medtech community’s initial concerns with the mdr’s requirements have now become a matter of increasing frustration for. The final texts of the new european medical devices regulation (mdr) and ivd regulation (ivdr) have been published in the official journal of the european union. Unlike the previous medical device directive, the mdr is directly applicable, without exception, in all european member states.
 Source: greensofttech.com
Source: greensofttech.com
The new regulation of medical devices aims to establish the path for medical device manufacturers to enter the european market. Usually it is the same person who the management representative as defined by iso 13485. Entry into force commenced on may 25th 2017, marking the start of the transition period for manufacturers selling medical devices into europe. However, in may 2021, the uk’s medical device laws deviated from those of the eu, due to the adoption of new regulations. The mdr, which replaces the medical.
If you find this site good, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title medical device regulation in europe by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.
Category
Related By Category
- Metastatic thyroid cancer prognosis
- Endocrinologist diabetes type 2
- How fast does colon cancer spread
- Hip replacement in elderly
- Physical therapy after arthroscopic shoulder surgery
- Symptoms of bacterial meningitis in children
- Chromophobe renal cell carcinoma
- Eye color change surgery usa
- Pradaxa vs eliquis vs xarelto
- Advanced stomach cancer symptoms